What Makes Water Wet?


Why is water wet? Its not just a question to boggle the minds of the kid scientists you encounter: Its a question of chemistry.
Nautilus recently posed just that question to Richard Saykally , a University of California, Berkeley chemistry professor who specializes in water, and here is how he explained what is going on:
When my daughters were very little we had an interesting revelation on that subject. I was actually giving both of my daughters a bath when they were very young and my youngest daughter said, Daddy? Why is water wet? And the proper answer is: strong tetrahedral hydrogen bonding, which they then related to their teachers for years afterward whenever the subject of water came, theyd say, Strong tetrahedral hydrogen bonding! But thats the correct answer. Thats what makes water wet.
Just the phrase strong tetrahedral hydrogen bonding may not mean much to you. It starts to mean a little more though when you think not just about how it fits together, but also the ways in which waters tetrahedral hydrogen bonds are unusual.
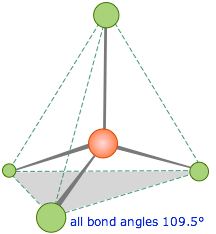
This right here is a standard tetrahedral bond. It looks as youd expect, a pyramid with the bonds all at a regular 109.5 away from each other. But water is a weird case in the world of tetrahedral bonding with its set of unpaired electrons pushing the two hydrogen molecules closer than theyd like to be to the tune of 104.5, making the whole big pyramid slope at a angle.
So when you hear chemists talk about how water has a bent structure thats what they mean. Saykally then goes on to explain how these bent and bonded water molecules clustering together makes up its larger structure:
A water cluster is an arrangement of two or more water molecules. So they adopt various structures. Two water molecules doesnt really have much of a shape; three water molecules makes a three-membered ring; four makes a squarish-looking ring; five makes a pentagon; and when you get to six water molecules, the morphology changes from being cyclic planar to being a three-dimensional cage; and thereafter seven, eight, nine and so on look like three-dimensional cages. The water eightthe eight-fold clusterlooks like a distorted cube, and then all larger clusters build on that cubic shape. These are the most stable forms that you would then find at very close to the absolute zero of temperature.
You can watch Saykallys whole interview where he also explains what the deal is with the 17 different kinds of ice that can form at Nautilus .
http://io9.com/what-makes-water-wet-1713082349






Science answers another of life's big questions.
And we learn an interesting factoid - who knew there were so many types of ice
The adaptive structure of the water molecules are responsible for the development of surface tension. Surface tension plays a big part in how water "feels". Just think about hard water and soft water.
Next up for you Robert, see if you can find an good article on the difference between stacked carbon atoms and randomized carbon atoms. Lets see if anyone takes notice.
Nice article BTW.....
FLY
I appreciate the perspective andintelligent feedback (unlike the peanut gallery input) on the subject.
We (at least I) do not always think about the complicated science behind the way that simple things just are.
How did they get less than eight molecules of water to happen ? Did they use a molecule counter ?
Petey
An excellent question for the professor, you should send your question to Berkley
17 different types of ice. My refrigerator has two, crushed and cube.
On a more serious note, I learned something today from the article. Thanks RIO
Kavika
You are most welcome
I enjoy a good science article now and again as well
Does the chemistry change when it is polluted?
To the poster of how to make hard boil eggs utilizing the wettest water...I apologize!
Great posting Robert.
I didn't think at first glance that this was a serious article, and then I read it and learned something I never knew before. Thanks, RIO, for posting articles that are sometimes entertaining, sometimes educational.
Buzz
You are welcome glad you liked it
O M
I would imagine it does since there would be other chemicals in the water that could (not positive) affect the chemical bonding and interaction of the molecules.
Lynne
Thanks for the feedback and those kind words - I am glad you liked the article.
FLY
Your point about hard and soft water, led me to the following explanation
I'm no chemist, but I think it could change with some pollutants and not others. " Oil and water don't mix ", comes to mind. Benzene should never be stored in or near your drinking water.
Sounds reasonable to me